Your Time has Finished
Loading...
BMAT 2009-S2
Your Score: %
Average Score of All Users:
You performed better than of students
Section Breakdown
| Your Score | Average of all Users | Percentile | |
|---|---|---|---|
| BMAT 2009 S2 |
BMAT 2009 S2
Your score:
Average score:
You performed better than of students
This section is Section 2 of 3.
Speed as well as accuracy is important in this section. Work quickly, or you might not finish the paper. There are no penalties for incorrect responses, only marks for correct answers, so you should attempt all 27 questions. Each question is worth one mark.
You must complete the answers within the time limit. Calculators are NOT permitted.
Good Luck!
Note – if press “End Exam” you can access full worked solutions for all past paper questions
Individual A in the family pedigree below is homozygous dominant and individual B is homozygous recessive for a particular feature.
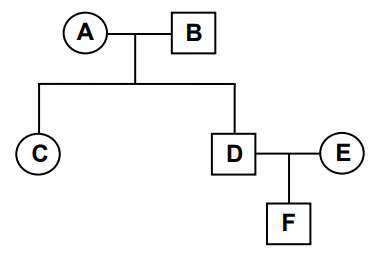
Explanation
The correct answer is C.
Let dominant allele = P, recessive allele = p.
A: homozygous dominant = PP; B: homozygous recessive = pp
| P | P | |
| P | Pp |
Pp |
|
p |
Pp |
Pp |
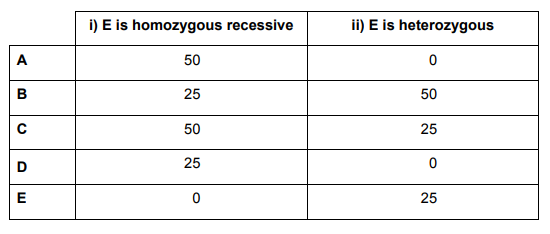
C and D are 100% heterozygous dominant. For F homozygous recessive:
- i) E is homozygous recessive:
E alleles = pp
| P | p | |
| p | Pp |
pp |
|
p |
Pp |
pp |
Ratio = 1 (PP) : 1 (pp)
Hence, there is a 50% probability that F is homozygous recessive
- ii) E heterozygous:
E alleles = Pp
| P | p | |
| P | PP |
Pp |
|
p |
Pp |
pp |
Ratio = 1 (PP) : 2 (Pp) : 1 (pp) – 1+2+1=4. pp = 1⁄4
Hence, there is a 25% probability that F is homozygous recessive
Which of the following could take part in an addition polymerisation?
1 CHI₃
2 C₂₄H₄₈
3 C₃H₇Br
4 C₄H₆Cl₂
5 C₈H₁₂Cl₄
Explanation
The correct answer is E.
Alkenes form addition polymers – the carbon-carbon double bond ‘opens up’ to form single carbon-carbon bonds with adjacent monomers. 1 and 3 are haloalkanes, so do not contain C=C bonds, and hence cannot form addition polymers. 2 is an alkene (as its molecular formula adheres to the general formula CnH2n) so is can under addition polymerisation. 4 and 5 are haloalkenes, so do have C=C bonds and can form addition polymers. So compounds 2, 4 and 5 can undergo addition polymerisation.
Shortly after opening her parachute, a free-fall parachutist of mass 60kg experiences the forces shown in the diagram.
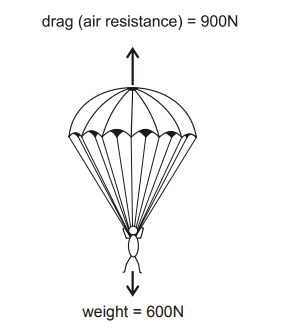
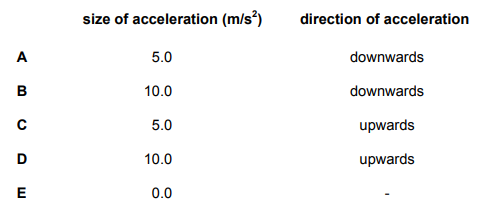
Explanation
The correct answer is C.
Resultant Force = 900N – 600N = 300N
Hence, force = upwards. a = F/m ; 300/60 = 5 ms-²
A bag contains x red balls, y blue balls and z yellow balls. One ball at random is taken out and replaced. A second ball at random is taken out and replaced.
Explanation
The correct answer is C.
The total number of balls: x+y+z
The probability of red: x/(x+y+z) , blue: y/(x+y+z) , yellow: z/(x+y+z)
Red first: x/(x+y+z) X blue second: y/(x+y+z) = 2xy/(x+y+z)²
Which one of the following statements is true about the members of a clone?
Explanation
The correct answer is E.
Cloning produces another organism, which is genetically identical to its parent organism – it contains the same genetic material.
Clones do not always show identical features at maturity, as the genes may be expressed differently as characteristics, dependent upon various transcription factors that dictate gene expression. Multiple births (I.e. twins, triplets etc.), are not always members of a clone. This is as, non-identical twins/triplets etc. can occur due to the release of multiple eggs at ovulation, which are subsequently fertilised by multiple sperms. The genetic material of the sperms and eggs differ from each other, due to genetic recombination (crossing over) in meiosis. Hence, they can express different characteristics. Members of a clone cannot be produced through a mutation; if this occurred, then their genetic material would differ immensely, and so they would be genetically non-identical – not members of a clone.
Which one of the following covalent substances could exist as a giant structure?
Explanation
The correct answer is B.
SiO₂ can exist as a giant structure – a covalent macromolecule, as it is held by strong covalent bonds. The strong covalent bonds link all the atoms together – a Si bonded to an O bonded to a Si bonded to an O etc.
Which of the following is a correct unit of potential difference (voltage)?
Explanation
The correct answer is E.
P = VI ; P = watts, I = amps. V = P/I. Hence, V = watts/amps = watts per amp
 koko
Medicmind Tutor
koko
Medicmind Tutor
Fri, 21 Jul 2023 13:24:47
why is V=IR not ccorrect
A cube has unit length sides. What is the length of a line joining a vertex to the midpoint of one of the opposite faces (the dashed line in the diagram below)?
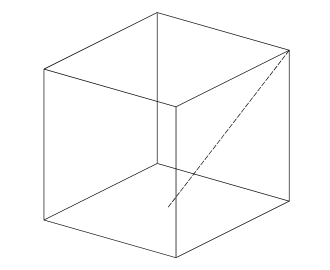
Explanation
The correct answer is B.
Unit length is defined as a length of 1. To find the diagonal of the base, use the Pythagorean Formula: √(1²+1²) = √2
The length of the midpoint of the diagonal: √2/2
Repeat usage of Pythagorean Formula: (the vertical height is 1, the base length to midpoint is √2, and the length in question is the hypotenuse of this triangle)
Hence, √((√2/2)²+1²) = √(1/2 + 1) = √(3/2)
The data below considers the risk of having a cancer in the neck region due to drinking alcohol and one genetic factor (presence of a mutant allele for the CYP1A1 gene). A risk value of 1.0 is the mean average risk in the human population.
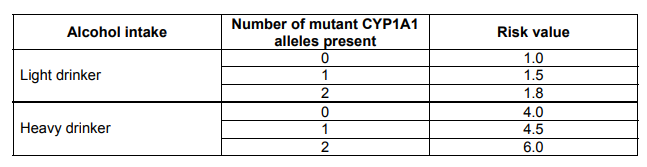
Explanation
The correct answer is D.
Statement D is incorrect. Statement A is correct, as the risk increases by 0.5 (1.0 ! 1.5 and 4.0 ! 4.5) in both types of alcohol intake. Statement B is correct, as there is a positive correlation of increase between the variables of number of mutant alleles present and risk value; as the number of mutant alleles present increases, so does the risk. Statement C is correct, as the figures for heavy drinking significantly increase the risk from being in the range of 1-1.8, to 4-6 which is an increase greater than fourfold. Statement E is correct, as the difference between the risk values with 1 and 2 alleles is 1.5 for heavy drinkers (6 – 4.5 = 1.5), whereas it is only 0.3 (1.8 – 1.5) for light drinkers. Statement D is incorrect, as the presence of at least one mutant allele is not the main factor for an increase in risk – this contradicts Statement C, which has proven that heavy drinking is the main factor which increases risk.
2.00g of a carbon compound is burnt in excess oxygen.
4.77g of carbon dioxide is produced.
Explanation
The correct answer is E.
Mr (CO₂) = 12 + 2(16) = 44
Ratio of C:Mass of CO₂ = 12/44 = 3/11
Hence, 4.77g of CO₂: 3/11 x 4.77 = 1.3g of C
For 2g of Carbon (Percentage by mass): Mass/Actual x 100 = 1.3/2 x 100 = 65%
It is known that a radioactive source emits a single type of radiation. Detectors are placed in the two positions shown. The graph shows how the readings change over time.
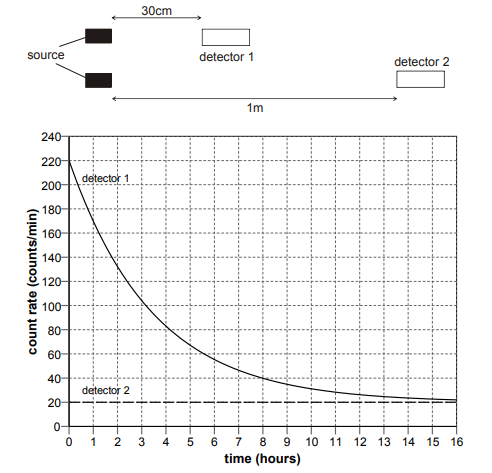
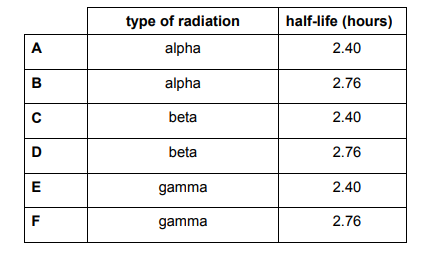
Explanation
The correct answer is C.
The radiation is Beta, as beta particles have a short range in air, a few cm, and a short half life of a few hours too – all values for half life are within the range of 2-3 hours. It cannot be alpha, as the range of alpha is too short – 3-5 cm. For half life: count rate initially = 220. Count rate halved at 110. Graphically, this corresponds to 2.40 hours – it cannot be 2.76 hours, as on the graph, the point is less than halfway. Detector 2 has a constant reading, as the range of beta particles is around 15cm, and so there is a constant rate of minimal beta particles being detected.
 Priya
Medicmind Tutor
Priya
Medicmind Tutor
Mon, 01 Nov 2021 21:41:28
The half-life I got is 2.76 as the point is more than halfway on the graph contrary to the explanation.
 Anas
Medicmind Tutor
Anas
Medicmind Tutor
Thu, 02 Dec 2021 18:58:37
Its more than half way ...... duuuuuuh!
What is the value of (2 ![]() 3)
3) ![]() 2?
2?
The symbol ![]() defines a mathematical binary operation such that
defines a mathematical binary operation such that 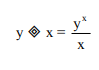 for all positive integers.
for all positive integers.
Explanation
The correct answer is C.
This question may appear to be confusing, However, this is just a substitution and calculation question. Firstly, identify that y X (mathematical binary operation) x = yᵡ/x
Let mathematical binary operation = ![]()
Hence, wherever that expression is seen, replace it with yᵡ/x, where x and y are defined.
For (2![]() 3)
3)![]() 2 Firstly, y = 2, x=3.
2 Firstly, y = 2, x=3.
Hence, 23/3 = 8/3
Then, y = 8/3, x=2
Hence, (8/3)²/ 2 = 64/9/2 = 32/9
Which one of the following movements will be severely and immediately disrupted if the oxygen concentration in the blood becomes very low?
Explanation
The correct answer is B.
If oxygen concentration in the blood becomes very low, all other processes, apart from B, are unaffected. This is as, processes A and C are gaseous exchange – if the blood oxygen concentration is low, oxygen can pass from the alveoli to the blood, down a concentration gradient. Similarly, the concentration of carbon dioxide in the blood will be high, due to the previous usage of oxygen in respiration to produce carbon dioxide, thus carbon dioxide also passes out of the blood, into the alveoli, down a concentration gradient. Water from the blood plasma passes into the muscle cells and nephron via osmosis, which does not require oxygen. Urea passes from the blood plasma into the nephron via diffusion, which does not require oxygen either. Only process B is affected. This is as, glucose enters the villi, from the lumen, via the glucose-sodium co-transport mechanism – facilitated diffusion. Here, glucose passes into the epithelial cells on the villi lining due to the presence of sodium ions. The sodium-potassium pump on the cell membrane of the epithelial cell bordering the capillary in the villi, actively transports sodium ions against their concentration gradient, out of the epithelial cells (and couples the transport of potassium ions into the cell). This causes a low concentration of sodium ions, promoting the entry of sodium ions, which couple with glucose, into the epithelial cell via the lumen. Due to a constant concentration gradient established by the active transport of sodium ions, glucose continuously moves out of the epithelial cells, into the villi capillaries. Active transport requires oxygen. If there is insufficient oxygen, the active transport of sodium cannot occur, hence sodium accumulates in the cell, stopping the concentration gradient of glucose and sodium into the cell.
The order of reactivity of four elements is J>M>Q>T i.e. J is the most reactive and T the least reactive.
A series of reactions are given below:
1 MCl₂ + T → TCl₂ + M
2 MSO₄ + J → JSO₄ + M
3 TO + Q → QO + T
4 QO + J → JO + Q
5 JSO₄ + T → TSO₄ + J
6 MO + Q → QO + M
Explanation
The correct answer is B.
MSO₄ + J → JSO₄ + M – Reaction 2 would occur, as J is more reactive than M, and so it would displace it from MSO₄.
TO + Q → QO + T – Reaction 3 would occur, as Q is more reactive than T, and so it would displace it from TO.
QO + J → JO + Q – Reaction 4 would occur, as J is more reactive than Q, and so it would displace it from QO.
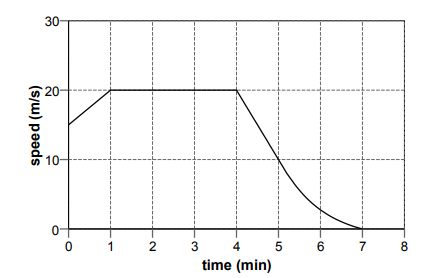
The graph represents the motion of a vehicle during part of a journey.
Explanation
The correct answer is D.
The trapezium (for 1 minute) can be calculated: (15 + (20×60)) x 1⁄2 = 607.5
Rectangle: (3×60) x (20) = 3600
Approximate area as triangle: (3×60) x 20/3 = 1200
Hence, 607.5 + 3600 + 1200 = 5407.5m nearest value is 6km
 Bob
Medicmind Tutor
Bob
Medicmind Tutor
Sun, 10 Oct 2021 22:24:43
Hi i think the area of a trapezium is ((a+b)/2 )x h .... so wouldn't it be (15+20)x60x1/2 =1050
 Kate
Medicmind Tutor
Kate
Medicmind Tutor
Sun, 25 Sep 2022 15:19:38
I agree with Bob
Evaluate:
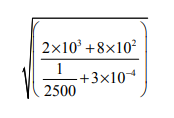
Explanation
The correct answer is F.
Simplify the expression
Which of the four statements below about natural selection are correct?
1 Competition occurs between individuals of the same species.
2 Competition occurs between individuals of different species.
3 Selection can lead to evolution.
4 Selection can lead to extinction.
Explanation
The correct answer is F.
All Statements 1,2,3 and 4 are correct. Statements 1 and 2 – Competition can occur between individuals of the same species (intraspecific competition) and between individuals of different species (interspecific competition). Statements 3 and 4 – Selection of advantageous alleles, which are favoured by the environment, can lead to evolution of a species, as they are passed onto successive generations. However, if a species does not possess any advantageous alleles to suit the changes in the environment, the individuals cannot survive to breed successfully, hence their alleles are not passed onto any offspring, leading to extinction.
The equation for the preparation of nitrogen monoxide is:
a Cu + b HNO₃ → a Cu (NO₃)₂ + c H₂O + 2NO
Explanation
The correct answer is D.
LHS: Cu = 1, H = 1, N = 1, O = 3. RHS: Cu = 1, H = 2, O = 8, N = 4
Using algebra:
Cu (1 x a = 1 x a) ; H (1 x b = 2 x c) ; N (1 x b = 2 xa+ 2);
O (3 x b = 6 x a +1 x c + 2)
Hence:
a=a
b=2c
b=2a+2
3b=6a + c + 2
We use substitution here:
6b=12a+2c+4
6b=12a+b+4
6(2a+2)=12a+(2a+2)+4
12a+12=12a+2a+2+4
12a+12=14a+6
2a=6
a=3
b=2a+2
b=6+2
b=8
b=2c
c=b/2
c=4
Hence, 3Cu + 8HNO₃ → 3Cu(NO₃)₂ + 4H₂O + 2NO
An object of mass 5kg falls from rest and hits the ground at a speed of 20m/s. Air resistance is negligible.
Take g to be 10m/s²
Explanation
The correct answer is B.
Loss of GPE = gain in Ek
mgh=1/2mv2 ; h = 1⁄2 (v2/g) = 1⁄2 x (20)2/10 = 20
A solid sphere of radius r fits inside a hollow cylinder. The cylinder has the same internal diameter and length as the diameter of the sphere.
The volume of a sphere is 4/3 π r³ , where r is the radius of the sphere.
Explanation
The correct answer is D.
The question says that the cylinder has the same length as the internal diameter of the sphere. Hence, h (length) = 2r for the cylinder
To find the fraction: Volume of sphere/Volume of cylinder
3/4πr³/ (πr²h)=3/4πr³/ (πr²(2r)) = 2/3
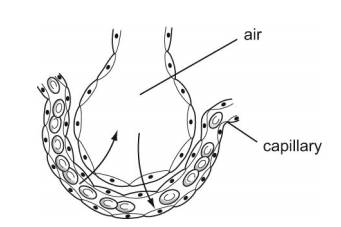
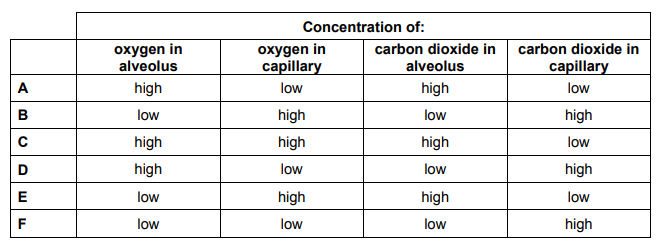
The diagram shows a section through an alveolus. Gases diffuse through the alveolar and capillary walls.
Explanation
The correct answer is D.
The blood in the capillaries is deoxygenated, with high carbon dioxide concentration. This is as oxygen is used by the cells for respiration, which produces carbon dioxide, which passes into the blood capillaries. This high concentration of carbon dioxide reaches the alveoli. Due to the air in the alveolus having a higher concentration of oxygen, relative to the capillaries, oxygen passes down a concentration gradient from the alveolus to the capillaries. The reverse occurs for carbon dioxide, as the blood capillaries has a greater carbon dioxide concentration, relative to the air in the alveolus. Hence, carbon dioxide moves out of the capillaries, into the alveolus, to be exhaled.
At room temperature, a reaction is very fast if no bonds are broken while it takes place, and is slow if bonds have to be broken.
1 H₂ + I₂ → 2HI
2 Ag⁺+ Cl⁻ → AgCl
3 ĊH₃ + ĊH₃ → C₂H₆
4 OH⁻+ CH₃Br → CH₃OH + Br⁻
Explanation
The correct answer is D.
Equation 2 is very fast, as it occurs between oppositely charged ions, which are attracted to each other due to electrostatic forces of attraction between them. Ag⁺ + Cl- rapidly combine AgCl.
Equation 3 is very fast as no bonds have to be broken. Also, the radicals combine rapidly, as they have a single unpaired electron in the outermost shell, as denoted by the dot. As both radicals have this, they combine to form a single C-C covalent bond, forming C₂H₆ (formed from .CH₃ + .CH₃).
Equations 1 and 2 are slow. Equation 1: H₂ needs to break up into 2 single H atoms, and I₂ needs to break up into 2 single I atoms. Then, 2HI molecules are formed separately. Hence, bonds are broken.
Equation 2: Firstly, there is no attraction to the negatively charged OH- ion, as the CH₃Br is uncharged. Secondly, via electrophilic substitution, the OH- needs to replace the Br=, which involves bond breaking.
A train consists of a powered engine pulling two unpowered carriages.
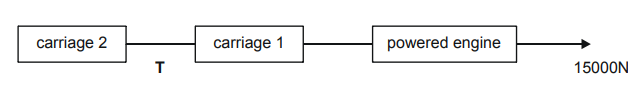
The engine has a mass of 20000kg, and each carriage has a mass of 5000kg. When the engine accelerates from rest it develops a thrust (driving force) of 15000N as shown.
Explanation
The correct answer is A.
F=ma
15000 = (20000+2(5000))a
a=1/2
15000 – T = (20000+5000)a
T = 15000 – 12500 = 2500
The equation gives y in terms of x
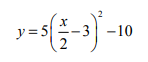
Explanation
The correct answer is A.
Add 10 to both sides: y + 10 = 5((x/2)-3)²
Divide by 5 on both sides: (y+10)/5 = ((x/2)-3)²
Square root both sides (remembering that there are positive and negative square roots): ± √((y+10)/5) = ((x/2)-3)
Add 3 to both sides: 3 ± √((y+10)/5) = x/2
Multiply by 2 on both sides: 6 ± 2(√((y+10)/5)) = x
The following five statements relate to homeostasis (the maintenance of stable internal conditions of the body):
1 Insulin helps to control the water content of the body.
2 Homeostasis depends only on hormones.
3 The pancreas helps to control the glucose content of the blood.
4 Both the nervous and hormonal systems are involved in homeostasis.
5 The skin is used to help control the body temperature.
Explanation
The correct answer is F.
Statement 1 is incorrect – insulin regulates blood glucose level, not water content. ADH regulates water content of the body.
Statement 2 is incorrect – Homeostasis is not only dependent on hormones – the nervous system can influence the release of hormones, such as the release of hormones noradrenaline and acetylcholine at the SAN to regulate heart rate, when detected by chemical and pressure changes in the blood flowing through the medulla oblongata in the brain, via the parasympathetic (decreases heart rate) and sympathetic (increases heart rate) nervous systems. Statements 3,4 and 5 are all correct. The pancreas produces insulin to control glucose content of the blood. Both nervous and hormonal systems are involved in homeostasis (which contradicts Statement 2), and thermoreceptors on the skin can help to control body temperature.
Naturally occurring chlorine is a mixture of two isotopes with mass number 35 and 37. The isotope with mass number 35 is three times as common as the isotope with mass number 37. Naturally occurring bromine is a mixture of two isotopes with mass numbers 79 and 81. They are present in equal amounts.
Explanation
The correct answer is C.
In CH2BrCl, the CH2 will be constant. Hence, Ar (CH2) = 12 + 2 = 14.
Hence, the variant part is: 128 – 14 = 114
To get 114, the only combination is: 35 + 79 = 104
There is 1⁄2 chance of 79 – Br isotope, and 3⁄4 chance of getting the 35 – Cl isotope.
As they are independent, multiply the fractions together, as they are probabilistic quantities: 1⁄2 x 3⁄4 = 3/8
The first graph shows the variation of the displacement of particles with distance along a wave at a particular instant in time:
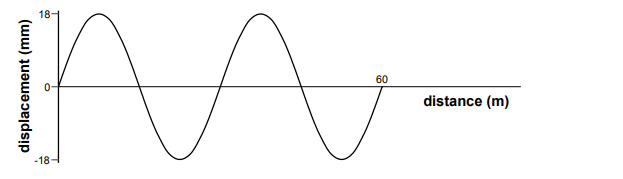
The second graph shows the variation with time of the displacement of a particular particle in this wave:
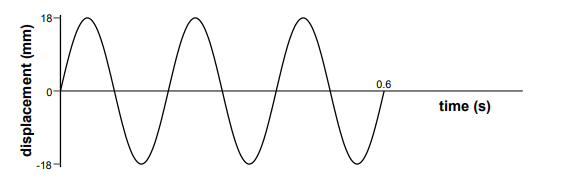
Explanation
The correct answer is E.
The displacement distance graph gives the wavelength of the wave. The wavelength is the distance between peak to peak, or trough to trough of a wave. However, only the total distance is given. There are 2 waves within that distance, and the distance is 60m. Hence, the wavelength = 60/2 = 30.
The displacement time graph gives the time period of the wave. The time of the waves is 0.6s, and there are 3 waves. Hence, the time period: 0.6/3 = 0.2s. The frequency is 1/time period. Hence, 1/0.2 = 5.
As c=f�, 30 x 5 = 150.
BMAT 2009 S2 Review Screen
Instructions
Below is a summary of your answers. You can review your questions in three (3) different ways.
The buttons in the lower right-hand corner correspond to these choices:
1. Review all of your questions and answers.
2. Review questions that are incomplete.
3. Review questions that are flagged for review. (Click the 'flag' icon to change the flag for review status.)
You may also click on a question number to link directly to its location in the exam.
BMAT 2009 S2 Section
Final Answer Review Screen
Instructions
This review section allows you to view the answers you made and see whether they were correct or not. Each question accessed from this screen has an 'Explain Answer' button in the top left hand side. By clicking on this you will obtain an explanation as to the correct answer.
At the bottom of this screen you can choose to 'Review All' answers, 'Review Incorrect' answers or 'Review Flagged' answers. Alternatively you can go to specific questions by opening up any of the sub-tests below.
BMAT 2009 S2 Section
TI-108